
A long-acting formulation of rifabutin is effective for prevention and treatment of Mycobacterium tuberculosis
Tuberculosis (TB) is a communicable disease caused by Mycobacterium tuberculosis (Mtb) and is a major cause of morbidity and mortality. Successful treatment requires strict adherence to drug regimens for prolonged periods of time. Long-acting (LA) delivery systems have the potential to improve adherence. Here, we show the development of LA injectable drug formulations of the anti-TB drug […]
Tuberculosis (TB) is a communicable disease caused by Mycobacterium tuberculosis (Mtb) and is a major cause of morbidity and mortality. Successful treatment requires strict adherence to drug regimens for prolonged periods of time. Long-acting (LA) delivery systems have the potential to improve adherence. Here, we show the development of LA injectable drug formulations of the anti-TB drug rifabutin made of biodegradable polymers and biocompatible solvents that solidifies after subcutaneous injection. Addition of amphiphilic compounds increases drug solubility, allowing to significantly increase formulation drug load. Solidified implants have organized microstructures that change with formulation composition. Higher drug load results in smaller pore size that alters implant erosion and allows sustained drug release. The translational relevance of these observations in BALB/c mice is demonstrated by (1) delivering high plasma drug concentrations for 16 weeks, (2) preventing acquisition of Mtb infection, and (3) clearing acute Mtb infection from the lung and other tissues.
Introduction
Tuberculosis (TB), which is caused by Mycobacterium tuberculosis (Mtb)1 is a world health concern with high morbidity and mortality2. An estimated 10 million people developed TB in 2020, resulting in 1.3 million deaths2. Moreover, approximately one-fourth of the world’s population has a latent TB infection (LTBI) with the potential for reactivation2. Of additional concern is the rise in drug-resistant Mtb2. Early diagnosis, treatment of all patients with TB, prevention of LTBI reactivation, and prevention of initial TB infection are all crucial to the WHO strategy in combating the TB epidemic3. TB preventative therapy with a single anti-TB drug, which acts to prevent initial infection or reactivation of LTBI, is highly effective and can reduce TB incidence when taken consistently4. For cases of active drug-susceptible TB disease, multidrug treatment regimens have an intensive phase of 2 months, followed by a continuation phase of at least 4 months5. Non-adherence to TB treatment can lead to treatment failure and the development of drug resistance6. Long-acting (LA) parenteral drug formulations that provide sustained drug release over weeks or months have the potential to reduce dosing frequency such that only one or two injections of the drug could be sufficient for TB treatment. This would dramatically change anti-TB treatment, as less frequent dosing could increase treatment compliance and consequently limit the occurrence of drug resistance7,8,9. LA bedaquiline for prevention of LTBI reactivation is currently being developed10,11. Affordable LA formulations with generic anti-TB drugs would allow the use of this approach in low-income communities.
LA biodegradable in situ forming implant (ISFI) formulations are attractive due to their unique properties which allow for subcutaneous administration of liquid formulations that solidify and form an implant at the site of injection. Injectable formulations are less invasive and less painful to administer than solid implants, and the biodegradable nature of the polymer matrix eliminates the need for surgical implant removal12. However, in the event of serious adverse reactions or at the completion of the treatment, the implant can be removed13,14. The removal of the implant stops drug delivery and prevents long-term exposure to subtherapeutic RFB concentrations13,14. To develop an ISFI LA formulation, the drug of interest and biodegradable polymer are solubilized in water miscible organic solvents12. Upon injection, phase transition occurs by solvent exchange, and polymer precipitation results in the formation of a solid implant consisting of biodegradable polymer and drug12,14. The drug release from the implant is controlled by implant structure15,16, polymer biodegradation17 and the composition of the liquid formulation18. Several ISFI formulations have been approved by the Food and Drug Administration (FDA) and include treatments for cancer, schizophrenia, and opioid dependency19,20,21,22,23,24. Eligard, Sublocade, and Perseris utilize biodegradable poly(lactic-co-glycolic) acid (PLGA) and N-methylpyrrolidone (NMP). Atridox contains biodegradable polylactic acid (PLA) and NMP. Onyx®Liquid Embolic system is a drug-free formulation based on ethylene vinyl alcohol co-polymer and dimethyl sulfoxide (DMSO) solvent. All formulations are injectable and form an implant after administration20,21,22,23,24.
Rifamycins, including rifampin (RIF), rifapentine (RFP), and rifabutin (RFB), are the cornerstones of TB therapy due to their potent bactericidal activity and their ability to inhibit DNA‐dependent RNA synthesis in prokaryotes25. RFB is a hydrophobic drug (LogP = 4.7) with reduced potential for drug-drug interactions, which makes it more suitable for treatment of Mtb/HIV coinfections compared to other rifamycins26,27. RFB also has higher tissue uptake due to its high lipophilicity, larger volume of distribution, longer terminal half-life, lower minimum inhibitory concentration (MIC) for Mtb, and higher tissue-to-plasma drug concentration ratio compared to RIF26,27,28,29. In humans, oral administration of 150 mg of RFB resulted in Cmax 460 ng mL−1, Cmin 50 ng mL−1, and tissue to plasma ratio of 5.6–6.8 in the lungs30,31. RFB is available as a low-cost generic medication and was selected as a model drug for development of a LA anti-TB drug formulation in this study. Multiple studies illustrate that RFB is safe and effective for treatment of active TB when patients have an adverse reaction to RIF, and RFB is particularly recommended instead of RIF for treatment of TB in patients with HIV utilizing certain antiretroviral therapies32,33,34.
ISFI LA formulations require a delicate composition balance to accommodate sufficient amounts of drug and polymer solubilized in biocompatible solvent. This severely limits the amount, number, and types of drugs that can be successfully formulated and represents a significant challenge when formulating RFB into an ISFI. Here, we show the development of LA-RFB injectable formulations that contain a biodegradable polymer (poly(lactic-co-glycolic-acid) (PLGA), a biocompatible water miscible solvent, RFB, and an amphiphilic additive (Fig. 1a, b) that dramatically increases drug load (Fig. 1c). After injection, these liquid formulations solidify in hydrophilic environments into an implant with porous microstructures, slow implant erosion and extended drug release (Fig. 1d, e). In vivo, a single subcutaneous injection of LA-RFB is able to deliver drug for 4 months, efficiently preventing Mtb infection after aerosol exposure, treating acute Mtb infection (Fig. 1f), and preventing Mtb dissemination to other organs.
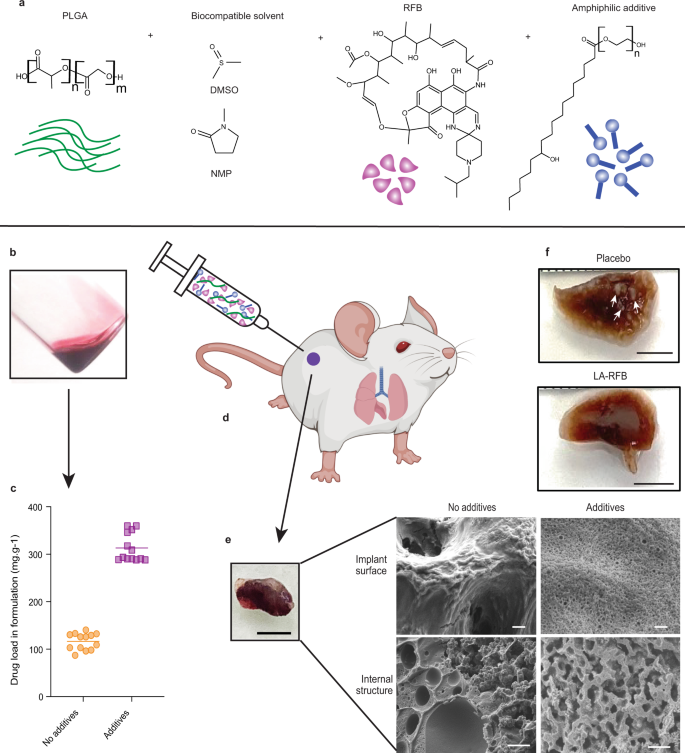
Results
Amphiphilic additives enhance RFB solubility and improve LA-RFB release kinetics
LA-RFB formulations were prepared by dissolving RFB at the maximum solubility in DMSO or NMP (202 ± 22 and 148 ± 34 mg mL−1, respectively, mean ± SD). Biodegradable ester-capped PLGA polymer with different molecular weights (MW, 10.6 kDa, 22.9 kDa, or 36.8 kDa and an LA:GA ratio of 50:50) was added to the RFB solution at multiple solvent to polymer mass ratios (2:1, 4:1, and 5:1) (Table 1). The resulting formulations were evaluated for (1) their injectability, (2) implant formation after injection to aqueous release medium (PBS), (3) initial release burst, defined as a release of RFB (%) within the first 72 h of incubation in release medium, and (4) daily release rate (μg day−1) at 37 °C with 100 rpm shaking under sink conditions (concentration of RFB in release medium <42 μg mL−1) (Table 1).Table 1 In vitro characteristics of LA-RFB formulationsFull size table
All 13 formulations were injectable, solidified after injection into PBS, and released RFB for over 8 weeks. Examples of release profiles and daily release rates are shown in Supplementary Fig. 1a, b. Release properties changed based on the composition of the individual formulation. Specifically, the initial release burst was higher in formulations with a 4:1 solvent to polymer ratio than in formulations with a 2:1 solvent to polymer ratio (Table 1). RFB daily release rates at 4 weeks of incubation were significantly higher in formulations with low MW PLGA compared to formulations with higher MW PLGA (Supplementary Fig. 1c). LA-RFB formulations with DMSO and a low MW PLGA (10.6 kDa) showed higher release rates at 4 weeks compared to formulations with NMP (Table 1, Supplementary Fig. 1d). These results demonstrate that by manipulating the composition of the ISFI formulation, release of drug from solidified implants can be modulated14.
Amphiphilic additives improve drug solubility in water by trapping molecules and forming nanoscale aggregates; micelles35,36. DMSO and NMP are polar solvents. The addition of Kolliphor®HS 15, an amphiphilic nonionic surfactant formerly known as Solutol HS 15, to DMSO dramatically increased RFB solubility (Fig. 2a). The highest RFB solubility, 564 ± 7 mg RFB per mL of DMSO (2.8 times higher than the RFB solubility in DMSO alone (202 ± 22 mg mL−1)) was achieved at 0.45% Kolliphor®HS 15. Similarly, the presence of Kolliphor®HS 15 in NMP also increased RFB solubility from 148 ± 34 mg mL−1 to 702 ± 10 mg mL−1 in NMP with 8.8% Kolliphor®HS 15 (Supplementary Fig. 2a). RFB solubility in DMSO was also substantially improved in the presence of low concentrations of other amphiphilic compounds, including D-α-Tocopherol polyethylene glycol 1000 succinate (TPGS, Supplementary Fig. 2b), Tween 20, Tween 80 (Supplementary Fig. 2c), and Pluronic F127 and F68 (Supplementary Fig. 2d).
